Using Lime in Organic Systems
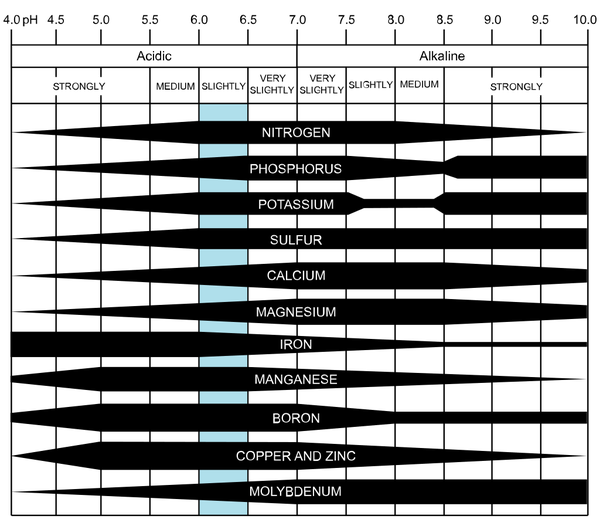
A soil’s ability to hold onto nutrients is termed cation exchange capacity (CEC), which describes the amount of negatively charged sites in a soil that can bind to positively charged nutrients. A low CEC means a soil does not hold on to nutrients well, and those nutrients may be lost through leaching. Having an optimal pH will improve the CEC of a soil and ultimately increase the availability of nutrients (Figure 1). When soil pH is too low, hydrogen and aluminum will out-compete important plant nutrients for the negatively charged sites in soil, making the nutrients unavailable to the plant. In managing your soil and interpreting soil health tests, CEC is an important factor for nutrient availability.
The pH of soil also impacts plant health. When soil has an optimum pH it encourages healthy root development and decreases plant stress. Having an ideal soil pH will also improve nitrogen fixation in legumes.Using Lime Effectively
‘Lime’ refers to a variety of products which are derived from limestone which neutralize acidity in soils. Limestone, either calcitic lime (CaCO3) or dolomitic lime (CaMgCO3) makes soil less acidic by reacting with hydrogen and aluminum cations.
Before applying lime, it is important to know the starting pH of your soil as well as the optimal pH for your crop (Figure 2). Not every crop will have the same pH tolerance and this will change based on your soil’s organic matter and clay content. It is important to consider your soil type when determining the ideal pH for your crop and the amount of lime you need to apply. Targeting lime applications for an optimal pH for your crop and soil will maximize its benefit and economic return.
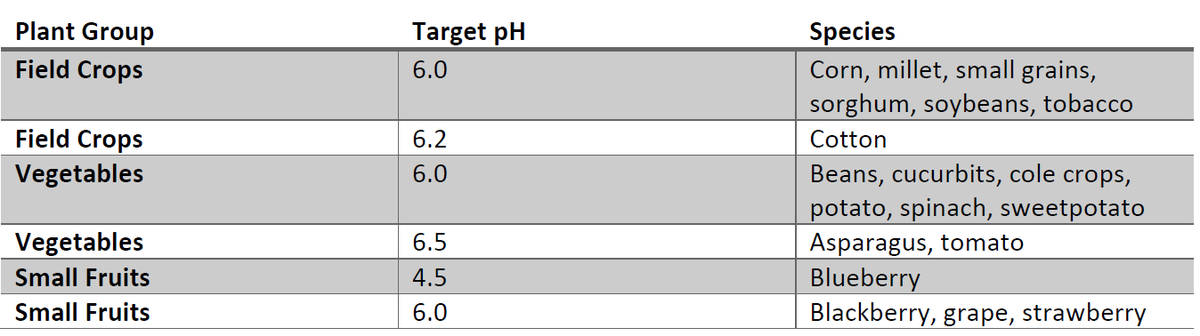
Table 1. Soil classification and target pH, Source: SoilFacts Soil Acidity and Liming for Agricultural Soils
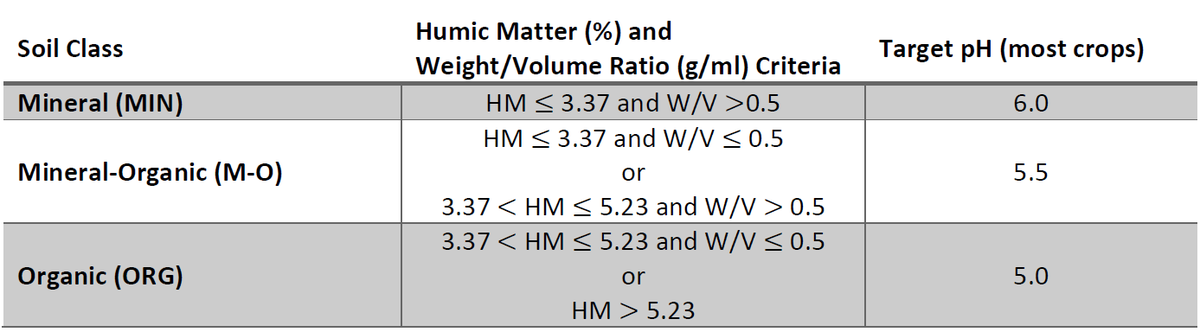
Table 2. Target pH based off crop, Source: SoilFacts Soil Acidity and Liming for Agricultural Soils
A routine soil test from NCDA will include soil pH and acidity, levels of major plant nutrients, micronutrients, sodium, percent base saturation (the percent of basic cations, as opposed to acidic Hydrogen and Aluminum cations, held on negative sites- an important factor for nutrient availability), percent of humic matter, Cation Exchange Capacity, and weight-to-volume ratio (soil density).
The NCDA calculates lime rates by comparing a soil’s current pH, target pH (based on the soil type and crop), the exchangeable acidity of soil (Ac), and in cases where lime was applied within the last year, a residual credit (RC) from the previous application (Figure 2). Exchangeable acidity (Ac) is a measure of the hydrogen and aluminum cations currently occupying the negative, cation exchange sites in the soil.
Residual Credit or RC is calculated by the rate of lime application (rate in tons/ac), how long ago it was applied (months), and the reaction rate of the lime (percentage based on soil type) (Figure 3). This will determine how much of the lime from the previous application is still available to react with hydrogen and aluminum cations.
![tons lime / acre = Ac[(target pH - current pH) / (6.6 - current pH)] - RC](https://eit-wagpress-prod.s3.amazonaws.com/media/images/2023-04-formula-for-lime-application-NCDA.max-1200x700.png)
Figure 2. Formula for lime application rate. Source: Understanding the Soil Test Report, NCDA
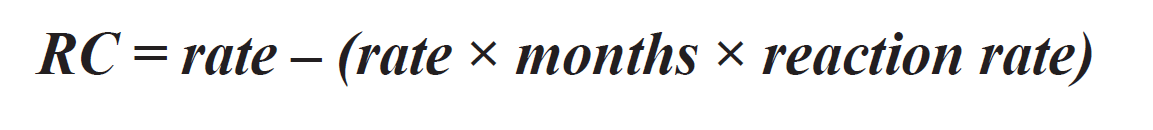
Figure 3. Formula for residual credit. Source: Understanding the Soil Test Report, NCDA
Based on these measurements, a soil test report will provide specific lime recommendations (Figure 4). If you list two crops, the recommendation will be for the crop that is least acid tolerant. Recommended lime rates are expressed as tons per acre in an agricultural setting.

Figure 4. Example soil test with different aspects labeled. Source: Know How to Use Your Soil Test Results, NC Soybean Producers Association
Targeting your lime application rate based on your current and target pH, crop, and soil type is the best way to ensure an economic return on the application. While organic producers are permitted to use lime, they are limited in what kind of lime they can use. It is important to check that the lime you are considering applying is compliant with the standards of the National Organic Program.
Organic Lime versus Non-Organic Lime
Wait…Isn’t lime natural? It’s limestone afterall…a rock

. Yes, liming products are sourced from limestone but some have undergone processing steps that make them not suitable for use in organic systems.
The National Organic Program allows only for the use of liming products that are in naturally mined forms for the purpose of managing soil fertility. Dolomitic and calcitic lime are considered naturally mined forms of limestone. Dolomitic and calcitic lime both bring soil pH up, the difference between them is that dolomitic lime has a significant amount of magnesium. When choosing between dolomitic and calcitic lime, consider if your soil has a magnesium deficiency.
Hydrated lime is not allowed for use as a means of bringing up soil pH. Burnt lime is also not permissible for use in organic systems. It is important to ensure that the lime you are applying is organically approved. There are many products to choose from which are listed by organic material review organizations, however if you are uncertain whether a product is compliant it is best to reach out to either your organic certifying agent or an extension agent.Additional Resources
For more information on soil acidity and lime, and how to calculate lime rates visit NC State’s SoilFacts publication Soil Acidity and Liming for Agricultural Soils
For more information on managing soil in organic system, visit the North Carolina Organic Commodities Production Guide chapter, Soil and Fertility Management
For more information on organic listed lime products with the Organic Material Review Institute (OMRI) review their approved organic inputs list
For more information on soil testing by NCDA, visit their Agronomic Services- Soil Testing page or check out their Understanding the Soil Test Report Publication